This blog post was inspired by Dr Marcia Philbin’s Speaker Series talk
What are polymers?
- A substance or material consisting of very large molecules, or macromolecules, composed of many repeating subunits.
- Their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life.
- Polymers range from familiar synthetic plastics such as polystyrene, to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function.
- They are joined together by strong covalent bonds, and have larger intermolecular forces compared to simple covalent molecules, therefore more energy is needed to break them down. This means that most are solid at room temperature, but they have lower boiling points than ionic or giant molecular compounds.

Image source: https://www.sciencenewsforstudents.org/article/explainer-what-are-polymers
How are polymers made?
Addition polymers:
The monomers that make up addition polymers have a double covalent bond between two carbons.
These monomers that have double bonds are unsaturated (alkenes), therefore they can open up their double bonds and join to form polymer chains. This is called addition polymerisation.
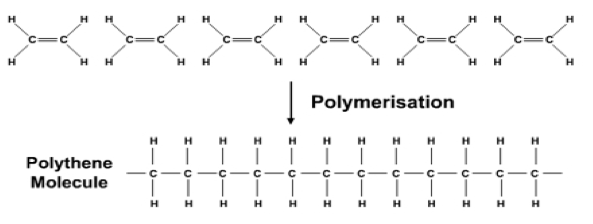
Image source: https://www.assignmentpoint.com/science/chemistry/addition-polymerisation.html
Condensation polymers:
Involves monomers which contain different functional groups, (e.g- alcohols, carboxylic acids, alkenes, etc.)
These functional groups react to form a polymer.
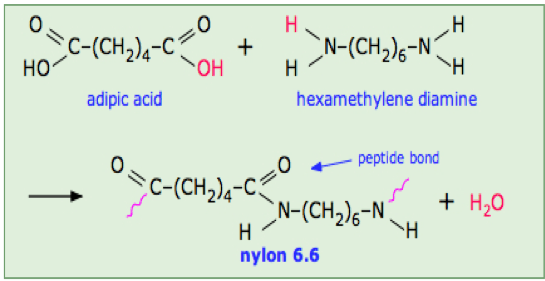
Image source: https://chem.libretexts.org/Courses/Sacramento_City_College/SCC%3A_CHEM_330_-_Adventures_in_Chemistry_(Alviar-Agnew)/10%3A_Polymers/10.05%3A_Condensation_Polymers
Examples of properties and uses of polymers
These are examples of polymers that are used in everyday life, but mostly go unnoticed. They play a vital part for many objects as they have desirable qualities and are overall accessible for producers. Polymers are even important in beauty products such as nail polish, for example it is used so it can create a film on the nail.
| Polymer | Properties | Uses |
| Poly(ethene) ‘polythene’ | Flexible, cheap, can be made into thin films | Carrier bags, shampoo bottles, food wrap |
| Poly(propene) ‘polypropylene’ | Flexible, strong, resists shattering | Buckets, bowls, crates, ropes, carpets |
| Poly(chloroethene) ‘PVC’ | Tough, electrical insulator, can be made hard or flexible | Insulation for electrical wires, windows, gutters, pipes |
| Poly(tetrafluoroethene) ‘PFTE’ | Slippery, chemically unreactive | Non-stick coatings for pans, containers for laboratory substances |
Advantages and disadvantages of polymers:
| Advantages | Disadvantages |
| Flexible | Chemical additives may be toxic if they leech into the environment |
| Malleable | When broken down, microplastics harm wildlife by polluting environment. |
| Strong | When burnt, it produces toxic fumes. |
| Inexpensive | It is costly to recycle |
| Lightweight | Non-biodegradable |
Potential uses of polymers in the future
Medicine: Both natural and synthetic polymers are used in medical prosthetic applications like heart valves, stents, cartilage scaffolds, joints, making of artificial skin, blood vessels, urinary catheters, ureteral stents, artificial kidney and also in nano systems for drug delivery. As the nano delivery of drugs to the target organs are an ongoing advancement, importance of biodegradable polymers in the field of medicine is sky rocketing. Biodegradability of materials used for nano delivery, both in the treatment and imaging, is important as the accumulation of used materials leads to toxicity in the body.
Polymers are currently being tested in the military sector to enhance the properties of defence mechanisms, such as helmets. For example, many governments are exploring how armour could be improved, and due to the polymer’s properties are investigating how to better protect soldiers and make armour less heavy. As polymers are lightweight, they can add a lot of strength to a material relative to its size and weight.
The potential use of polymer materials for devices has been investigated intensely over the past few decades. In particular, conjugated polymers show semiconductor-like behaviour and have emerged as intriguing materials for the fabrication of flexible, large-area, and low power and low-cost electronic devices. Moreover, the high absorption coefficients of polymers and polymer composites and the possibility of varying the band gap through molecular engineering have opened up new options for multicolour electrochromic devices and solar-energy conversion.
Thank you for reading and hope you enjoyed it!
Written by Malick, a Year 11 Student at Haberdashers’ Aske’s Knights Academy